In the fast-evolving landscape of precision medicine, the path from identifying a genetic mutation to deploying a clinical diagnostic tool is complex. The most critical milestone in this journey is biomarker validation. While prospective clinical trials are the “gold standard,” they are often sidelined by high costs and decade-long timelines.
The most efficient solution? Retrospective validation using FFPE (Formalin-Fixed Paraffin-Embedded) archived tissue samples. By analyzing preserved specimens with established clinical histories, researchers can confirm a biomarker’s predictive power in a fraction of the time.
- The Strategic Advantage of Retrospective Studies
Biomarker validation proves that a specific biological marker—be it a protein, gene mutation, or RNA pattern—is a reliable predictor of patient drug response or disease state.
The “Time-Machine” Effect
Using FFPE archives allows researchers to “look into the future” by studying the past.
- Prospective Studies:Require years of waiting to observe patient survival and drug efficacy.
- Retrospective FFPE Studies:Researchers select tissue from patients treated years ago. Because the clinical outcome is already known, the correlation between the biomarker and long-term survival is immediate.
Statistical Power through Global Biobanking
For rare cancers like Cholangiocarcinoma, a single hospital may only see a handful of cases. Partnering with a global network like ibiospecimen.com allows researchers to aggregate hundreds of cases, providing the statistical depth required for FDA and EMA regulatory approval.
- Overcoming Technical Barriers in FFPE Samples
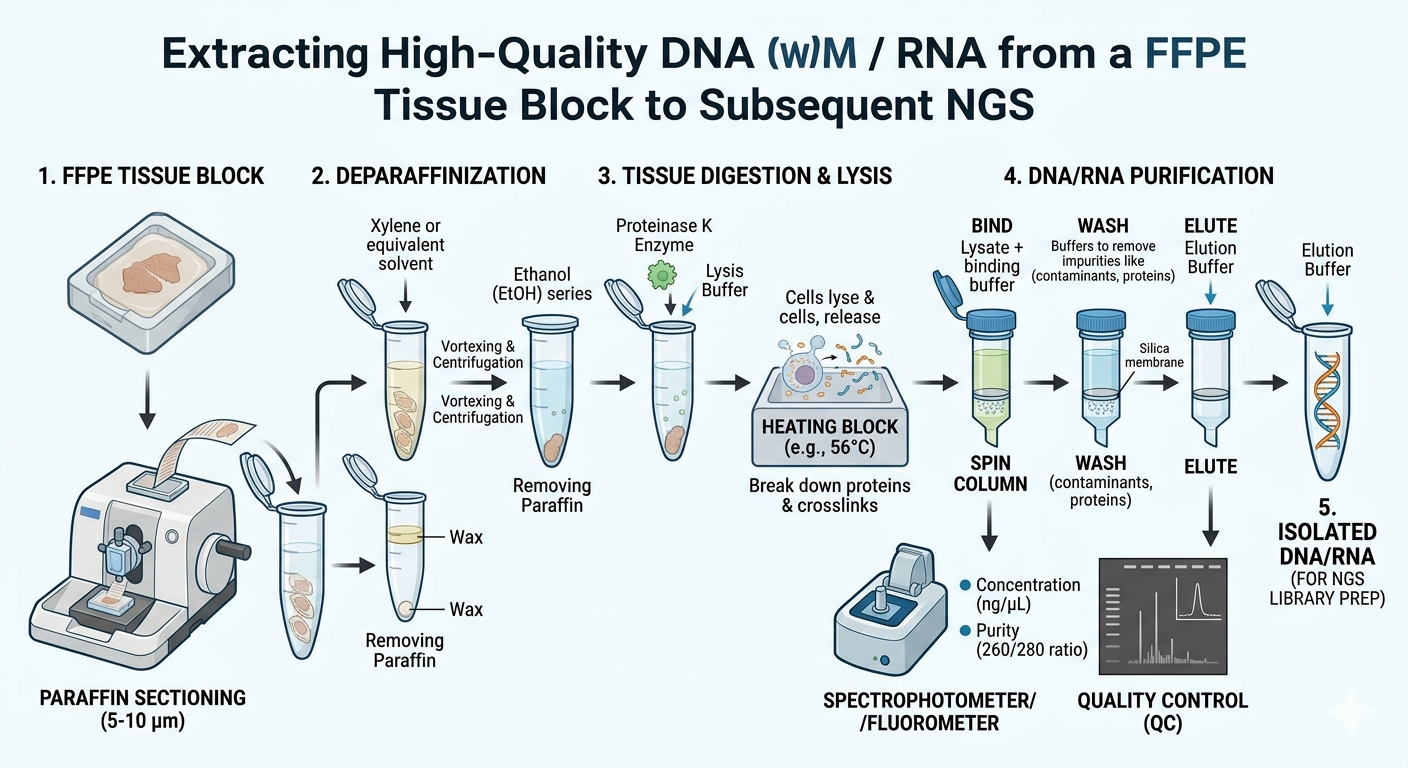
Validating biomarkers on FFPE tissue requires navigating the chemical changes caused by formalin, which can fragment nucleic acids and cross-link proteins.
- DNA-Based Biomarkers
DNA remains remarkably stable in paraffin. While High-depth Next-Generation Sequencing (NGS) is the standard, researchers must use advanced bioinformatics to filter out “C-to-T” transition artifacts caused by fixation.
- RNA-Based Biomarkers
RNA is prone to fragmentation. Experts utilize the DV200 score (percentage of RNA fragments >200 nucleotides) to ensure quality. Technologies like probe-based capture (e.g., Illumina RNA Exome) are then used to extract clear signals from fragmented “noise.”
III. Protein-Based Biomarkers (IHC)
Most clinical markers (PD-L1, HER2) are proteins. Antigen Retrieval (AR) is used to “unlock” the formalin cross-links, allowing antibodies to bind to epitopes for accurate staining intensity and concentration analysis.
- The Two Gates: Analytical vs. Clinical Validation
To reach the clinic, a biomarker must pass two rigorous phases:
| Validation Phase | Focus | Key Question |
| Analytical | Accuracy & Reliability | Does the test consistently detect the marker across different tissue lots? |
| Clinical | Medical Relevance | Does the marker actually predict patient survival or drug response? |
- Quality Control: Ensuring “Research-Ready” Samples
At ibiospecimen.com, we adhere to a “Quality-First” protocol because a validation study is only as good as its tissue source.
- Cold Ischemia Time (CIT):We prioritize samples with a documented CIT of <60 minutes to prevent protein degradation.
- Tumor Nuclei Percentage (TNP):To avoid “diluting” the genomic signal, validation cohorts typically require 30-50% tumor purity.
- Deep Clinical Annotation:Every block is linked to a de-identified dataset including treatment history, progression-free survival (PFS), and patient demographics.
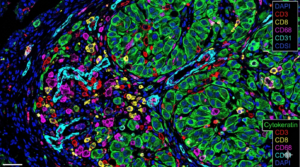
- The Future: Spatial Biomarkers & AI
In 2026, the focus has shifted from if a biomarker exists to where it is located. Spatial Transcriptomics and Multiplex Immunofluorescence (mIF) on FFPE sections allow researchers to see if immune cells (like CD8+ T-cells) are actually infiltrating the tumor core. This “Spatial Intelligence” is the new frontier of immunotherapy research.
- Regulatory Excellence and Ethics
When submitting data to the FDA, the chain of custody must be flawless.
- Compliance:All samples are fully de-identified and compliant with HIPAA and GDPR.
- Informed Consent:We ensure every donor has consented to commercial research and genomic sequencing.
- Transparency:Each specimen includes a Certificate of Analysis (CoA) for seamless regulatory bridging.
Why Choose ibiospecimen.com for Your Validation Study?
- Global Searchable Inventory:Filter thousands of blocks by mutation, IHC score, or survival data.
- Prospective Collection:If we don’t have it, we can custom-collect samples to your specific SOPs.
- Digital Pathology:Every block comes with a 40x Whole Slide Image (WSI) for AI-driven computational analysis.
Transforming Archives into Answers. The billions of FFPE blocks in storage are the keys to the next generation of cancer therapy. Let us help you unlock them.
Frequently Asked Questions
Q: How old can FFPE blocks be for validation?
For DNA and IHC, blocks can be up to 20 years old. For RNA signatures, we recommend blocks under 10 years old to maintain transcriptomic integrity.
Q: Can you help with rare biomarkers like NTRK or RET?
Yes. By leveraging our global network of surgical centers, we can aggregate rare cohorts that would take a single hospital years to collect.
Q: Do you provide matched primary and metastatic pairs?
Yes, matched pairs are available and are essential for validating biomarkers intended for late-stage or metastatic disease.
Ready to start your biomarker validation study?
Contact the specialists at ibiospecimen.com for a customized inventory report.
Email: info@ibiospecimen.com
